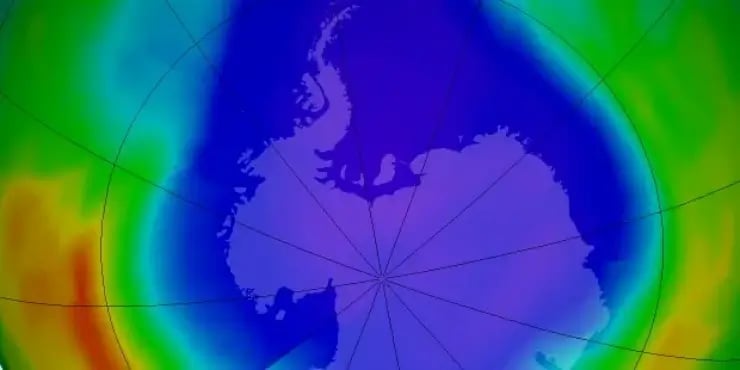
Two months ago, newspapers around the world happily reproduced a news headline from Reuters – the ozone hole was shrinking, it said, for the first time ever. The headline followed an announcement by U.N. Secretary-General Ban Ki-moon, who was celebrating the 25th anniversary of the Montreal Protocol, the historic agreement to ban ozone-destroying CFCs globally. How significant is the ozone layer recovery really? Do we have a reason to celebrate? Aren’t the CFC-replacing HFCs still a cause for concern, since they don’t destroy the ozone layer but do accelerate climate change? Can the Montreal Protocol teach us how to fight global warming?
The Two Faces of Ozone
For a complete understanding of the ozone hole, some basic knowledge of the chemical processes behind it comes in handy – chemists might kindly pardon me for the following simplifications and just skip this paragraph. But you should know that ozone is three connected oxygen atoms (O3). Oxygen exists in three forms: O (e.g. as bonded in H2O), O2 and O3. O2 is stable, whereas O3 (ozone) is highly reactive. Its extra O seeks a bonding partner to form a molecule like H20. The remaining O2 then leads a more stable life than the wild O3. Ozone plays two roles for living beings: a negative one near the earth’s surface, and a positive one in the stratosphere. The former, “ground level ozone”, can destroy the cellular structure of organisms (yes, because that third O is always on the run…) and is mostly derived from oxides of nitrogen (NOx) and volatile organic compounds (VOC) breaking down in the presence of sunlight. NOx and VOC are mostly byproducts of industry and automobiles. Ozone is also generated by lightning, artificial electrical sparks and photocopiers. Stratospheric ozone, plays a different role. It actually protects cellular structures indirectly because it “blocks” the vast majority of the sun’s ultraviolet light. The atmosphere, of course, has different layers. One in particular, the stratosphere, has the perfect conditions for ozone to accumulate. This is the layer where much of the ultraviolet light is blocked. Just before light from the sun finishes its way through space towards Earth, it hits the ozone layer. There, UV-rays split O3 into O and O2, thereby losing its energy. The released O soon finds another free O and forms another O2. This works well because the amount of “destroyed” O3 (by splitting) is matched by the amount of newly generated O3. However, when chlorofluorocarbon (CFC, the chemicals banned by the Montreal protocol) makes its way from a spray can to the stratosphere, the balance gets out of kilter. At ground level, CFCs are very stable and retain their molecular structure a long time. At the altitude of the ozone layer, however, the sun turns the CFCs into highly reactive little bastards: CFCs break apart under ultraviolet rays thus creating compounds that react with ozone. In a short time, more ozone is destroyed than nature can generate.
What the mass media articles often forget to mention, is that the Antarctic ozone hole, which reaches its annual peak every September, was indeed smaller this year than last year. However, in 2012, it was still bigger than two years ago.
How significant is the ozone layer recovery really?
First of all, we have to differentiate between CFC concentration on the one hand and the amount of ozone in the stratosphere on the other. Granted, seeing CFC concentrations in the atmosphere decrease is good news. However, that does not automatically mean the ozone layer recovers at the same pace. Second, we have to make another differentiation: the ozone layer has different dimensions in different parts of the world. Ozone floats in the atmosphere and is mobile. Consequently, we have to keep in mind geographical location, season and weather conditions when talking about ozone layer recovery. April Flowers, a redOrbit.com author, has published an excellent article on the Antarctic ozone hole recovery that puts the latest developments into perspective:
Despite the success of the [Montreal] Protocol for cutting production and consumption of these chemicals [chlorofluorocarbons], the chemicals have a very long atmospheric lifetime. It will take several decades before their concentrations are back to pre-1980 levels. The chemical saturation of Antarctic stratosphere reached a maximum around 2000 and is now decreasing at a rate of about 1% per year. […] Over the past ten years, stratospheric ozone in the Arctic and Antarctic regions is no longer decreasing, but it has not yet started to recover. The ozone layer outside the Polar regions is projected to recover to its pre-1980 levels before the middle of this century. However, the ozone layer over the Antarctic is predicted to recover around 2040. On the other hand, because of the impact of greenhouse gas warming, the ozone layer over the tropics and mid-southern latitudes may not recover for more than a century, and perhaps not ever.
Do we have a reason to celebrate?
The same article makes clear why CFC concentration and ozone hole size do not correlate directly:
Year-to-year variations in area and depth are caused by variations in stratospheric temperature and circulation. Colder conditions result in a larger area and lower ozone values in the center of the hole.
My apologies go to the celebration organizers; I didn’t mean to be a kill-joy. However, for your private ozone layer recovery fiesta, please make sure the beer is cooled in a HFC-free fridge. Which leads us to the next point:
Aren’t the CFC-replacing HFCs still a cause for concern?
Yes they are. HFC, also HCFC, stands for hydro-chlorofluorocarbon, as opposed to CFC, chlorofluorocarbon. On the positive side, we have to acknowledge that the presence of the hydrogen part of HFC takes out the danger of the CFC’s ozone-depleting nature. Nevertheless, the 20th-century’s ozone hole is global warming and, of all the substances that have replaced 98% of the CFCs in the last 25 years, it is HFCs that have the highest global warming potential.
Can the Montreal Protocol teach us how to fight global warming?
David Doniger, who was involved in the political action process in the US after the discovery of the ozone hole 40 years ago, hopes so. In his blog at the “Natural Resources Defense Council”, he stated:
Wisely, the Montreal treaty gives the parties the responsibility to ensure that replacement chemicals are safe – and that includes ensuring that they don’t magnifying climate change. So two groups of countries – the U.S., Canada, and Mexico, and a group of island nations led by Micronesia – have proposed using the Montreal Protocol to phase down HFCs. The vast majority of developed and developing countries want to move forward on this, but currently three countries – China, India, and Brazil – are blocking the start of negotiations. […] Despite the current stand-off on HFCs, the Montreal Protocol is proof positive that the earth’s nearly 200 countries can effectively cooperate to protect their citizens from a planetary pollution crisis.
We saved the ozone layer. We can save the climate.
Article image by NASA Ozone Watch, ozonewatch.gsfc.nasa.gov. It shows the antarctic ozone hole on September 22, when the 2012’s highest ozone hole area was reached (21 Mio km²).